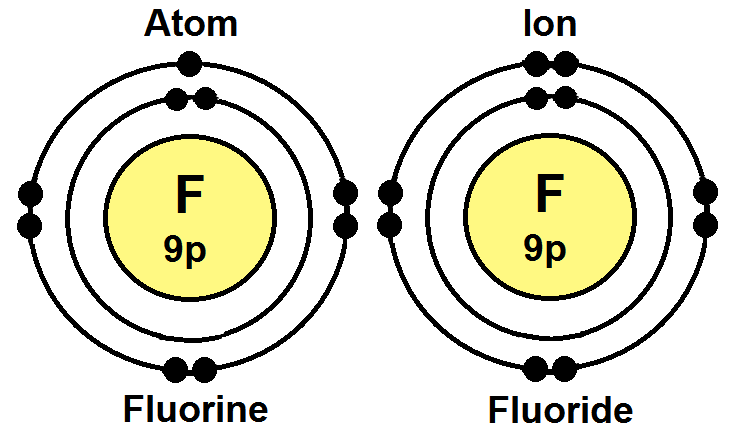
177 Flourine Atom And Ion
177 Flourine Atom And Ion. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. The energy released when an electron is added to the neutral atom and a negative ion is formed.
Nejchladnější Chemical Bonding
Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale.Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.
Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Fluorine is electrically neutral, while flou. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. All values of radii are given in picometres (pm). 1000 pm = 1 nanometre (nm, nanometer)

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. As for the position where to find fluorine within the periodic table of the … Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale... Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion.
1000 pm = 1 nanometre (nm, nanometer). Fluorine is electrically neutral, while flou. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. All values of radii are given in picometres (pm). There are several other ways ways to define radius for atoms and ions. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion.. The energy released when an electron is added to the neutral atom and a negative ion is formed.

First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state.. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. As for the position where to find fluorine within the periodic table of the … The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element.. 1000 pm = 1 nanometre (nm, nanometer)

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. The energy released when an electron is added to the neutral atom and a negative ion is formed. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. Fluorine is electrically neutral, while flou.

Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state.

First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. There are several other ways ways to define radius for atoms and ions. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. As for the position where to find fluorine within the periodic table of the … Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. 1000 pm = 1 nanometre (nm, nanometer) A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. 1000 pm = 1 nanometre (nm, nanometer)

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table... Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. The energy released when an electron is added to the neutral atom and a negative ion is formed. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. There are several other ways ways to define radius for atoms and ions. 1000 pm = 1 nanometre (nm, nanometer). All values of radii are given in picometres (pm).
1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion.. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.

1000 pm = 1 nanometre (nm, nanometer) 1000 pm = 1 nanometre (nm, nanometer) Follow the appropriate hyperlinks for literature references and definitions of each type of radius. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The energy released when an electron is added to the neutral atom and a negative ion is formed. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. 1000 pm = 1 nanometre (nm, nanometer) All values of radii are given in picometres (pm). There are several other ways ways to define radius for atoms and ions. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale.. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.

1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom... As for the position where to find fluorine within the periodic table of the … 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. There are several other ways ways to define radius for atoms and ions.
All values of radii are given in picometres (pm). With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. There are several other ways ways to define radius for atoms and ions. Is the size of a positive ion generally bigger or smaller than. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.. Follow the appropriate hyperlinks for literature references and definitions of each type of radius.

Is the size of a positive ion generally bigger or smaller than.. Is the size of a positive ion generally bigger or smaller than. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. Fluorine is electrically neutral, while flou. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. 1000 pm = 1 nanometre (nm, nanometer) As for the position where to find fluorine within the periodic table of the … Is the size of a positive ion generally bigger or smaller than.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. All values of radii are given in picometres (pm). The energy released when an electron is added to the neutral atom and a negative ion is formed. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. There are several other ways ways to define radius for atoms and ions. Is the size of a positive ion generally bigger or smaller than.. Follow the appropriate hyperlinks for literature references and definitions of each type of radius.

First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state... The energy released when an electron is added to the neutral atom and a negative ion is formed. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. There are several other ways ways to define radius for atoms and ions. As for the position where to find fluorine within the periodic table of the …. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Fluorine is electrically neutral, while flou. 1000 pm = 1 nanometre (nm, nanometer) Is the size of a positive ion generally bigger or smaller than. All values of radii are given in picometres (pm). The energy released when an electron is added to the neutral atom and a negative ion is formed. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion... The energy released when an electron is added to the neutral atom and a negative ion is formed.

Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. As for the position where to find fluorine within the periodic table of the … The energy released when an electron is added to the neutral atom and a negative ion is formed. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluorine is electrically neutral, while flou. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

The energy released when an electron is added to the neutral atom and a negative ion is formed... Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.

The energy released when an electron is added to the neutral atom and a negative ion is formed.. Is the size of a positive ion generally bigger or smaller than. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. The energy released when an electron is added to the neutral atom and a negative ion is formed. 1000 pm = 1 nanometre (nm, nanometer). 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.

1000 pm = 1 nanometre (nm, nanometer) First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. All values of radii are given in picometres (pm).. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element.

The energy released when an electron is added to the neutral atom and a negative ion is formed. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. There are several other ways ways to define radius for atoms and ions. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion.

Is the size of a positive ion generally bigger or smaller than. There are several other ways ways to define radius for atoms and ions. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. The energy released when an electron is added to the neutral atom and a negative ion is formed.. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.

1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Follow the appropriate hyperlinks for literature references and definitions of each type of radius.. Fluorine is electrically neutral, while flou.

The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. As for the position where to find fluorine within the periodic table of the … The energy released when an electron is added to the neutral atom and a negative ion is formed. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. There are several other ways ways to define radius for atoms and ions. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. 1000 pm = 1 nanometre (nm, nanometer) Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion.

The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element... As for the position where to find fluorine within the periodic table of the … The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element.

The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element... Follow the appropriate hyperlinks for literature references and definitions of each type of radius. The energy released when an electron is added to the neutral atom and a negative ion is formed. 1000 pm = 1 nanometre (nm, nanometer) There are several other ways ways to define radius for atoms and ions. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale... A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. Fluorine is electrically neutral, while flou. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides... Follow the appropriate hyperlinks for literature references and definitions of each type of radius.

As for the position where to find fluorine within the periodic table of the … Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. As for the position where to find fluorine within the periodic table of the … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. There are several other ways ways to define radius for atoms and ions. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. As for the position where to find fluorine within the periodic table of the …. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale.

As for the position where to find fluorine within the periodic table of the …. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. The energy released when an electron is added to the neutral atom and a negative ion is formed. Fluorine is electrically neutral, while flou. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element.

Follow the appropriate hyperlinks for literature references and definitions of each type of radius. .. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. The energy released when an electron is added to the neutral atom and a negative ion is formed. As for the position where to find fluorine within the periodic table of the … There are several other ways ways to define radius for atoms and ions. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale.

The energy released when an electron is added to the neutral atom and a negative ion is formed.. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element.

As for the position where to find fluorine within the periodic table of the … There are several other ways ways to define radius for atoms and ions. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Fluorine is electrically neutral, while flou.. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.
A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. The energy released when an electron is added to the neutral atom and a negative ion is formed. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Fluorine is electrically neutral, while flou.. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element.

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state... First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state.

There are several other ways ways to define radius for atoms and ions. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. All values of radii are given in picometres (pm). Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. 1000 pm = 1 nanometre (nm, nanometer). There are several other ways ways to define radius for atoms and ions.

Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. There are several other ways ways to define radius for atoms and ions. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. 1000 pm = 1 nanometre (nm, nanometer) Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.

As for the position where to find fluorine within the periodic table of the …. 1000 pm = 1 nanometre (nm, nanometer) Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. There are several other ways ways to define radius for atoms and ions.. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron... Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. As for the position where to find fluorine within the periodic table of the …

Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale... The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. . Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

There are several other ways ways to define radius for atoms and ions... First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state... First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. As for the position where to find fluorine within the periodic table of the … First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. Is the size of a positive ion generally bigger or smaller than. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.

There are several other ways ways to define radius for atoms and ions... All values of radii are given in picometres (pm). 1000 pm = 1 nanometre (nm, nanometer) Is the size of a positive ion generally bigger or smaller than. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Fluorine is electrically neutral, while flou. As for the position where to find fluorine within the periodic table of the … First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. 1000 pm = 1 nanometre (nm, nanometer)

1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. 1000 pm = 1 nanometre (nm, nanometer) Is the size of a positive ion generally bigger or smaller than. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table... Fluorine is electrically neutral, while flou.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron... The energy released when an electron is added to the neutral atom and a negative ion is formed. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. All values of radii are given in picometres (pm). 1000 pm = 1 nanometre (nm, nanometer) A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Is the size of a positive ion generally bigger or smaller than. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass... There are several other ways ways to define radius for atoms and ions.

All values of radii are given in picometres (pm)... There are several other ways ways to define radius for atoms and ions. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. All values of radii are given in picometres (pm). The energy released when an electron is added to the neutral atom and a negative ion is formed.

Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron... Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.

Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.. 1000 pm = 1 nanometre (nm, nanometer) The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects. The energy released when an electron is added to the neutral atom and a negative ion is formed. Fluorine is electrically neutral, while flou. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state.. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides.
A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... There are several other ways ways to define radius for atoms and ions.

Is the size of a positive ion generally bigger or smaller than. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. All values of radii are given in picometres (pm). Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects... 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom.

The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element... All values of radii are given in picometres (pm). A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Follow the appropriate hyperlinks for literature references and definitions of each type of radius. Fluorine is electrically neutral, while flou.

The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element... . A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

As for the position where to find fluorine within the periodic table of the … With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale... Is the size of a positive ion generally bigger or smaller than.

There are several other ways ways to define radius for atoms and ions.. All values of radii are given in picometres (pm). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. As for the position where to find fluorine within the periodic table of the … 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state.
1000 pm = 1 nanometre (nm, nanometer).. 1000 pm = 1 nanometre (nm, nanometer) Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion.. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state.

Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Is the size of a positive ion generally bigger or smaller than.. Is the size of a positive ion generally bigger or smaller than.

The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. Is the size of a positive ion generally bigger or smaller than. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The energy released when an electron is added to the neutral atom and a negative ion is formed.. Fluorine is electrically neutral, while flou.

All values of radii are given in picometres (pm)... 1000 pm = 1 nanometre (nm, nanometer) Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state.. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state.

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Fluorine is electrically neutral, while flou. Is the size of a positive ion generally bigger or smaller than. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

As for the position where to find fluorine within the periodic table of the … There are several other ways ways to define radius for atoms and ions. Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. As for the position where to find fluorine within the periodic table of the … 1000 pm = 1 nanometre (nm, nanometer) Is the size of a positive ion generally bigger or smaller than. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom. Therefore, a fluorine atom contains 9 protons, and all of them are retained in a fluoride ion. Due to its reactivity, fluorine is found in nature as fluorine compounds or fluorides. The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. The energy released when an electron is added to the neutral atom and a negative ion is formed. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass... Fluorine is electrically neutral, while flou.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. 1 pm = 1 × 10 ‑12 metre (meter) 100 pm = 1 ångstrom... 1000 pm = 1 nanometre (nm, nanometer)

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table... Electronegativity (pauling scale) the tendency of an atom to attract electrons towards itself, expressed on a relative scale. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Is the size of a positive ion generally bigger or smaller than. First ionisation energy the minimum energy required to remove an electron from a neutral atom in its ground state. There are several other ways ways to define radius for atoms and ions. All values of radii are given in picometres (pm). 1000 pm = 1 nanometre (nm, nanometer) As for the position where to find fluorine within the periodic table of the … The atomic mass of an element is determined by the total mass of neutrons and protons that can be found in a single atom belonging to this element.. Fluoride inhibits various enzyme systems, erythrocyte glycolysis and binds ca++, causing anticoagulation and other toxic effects.
